 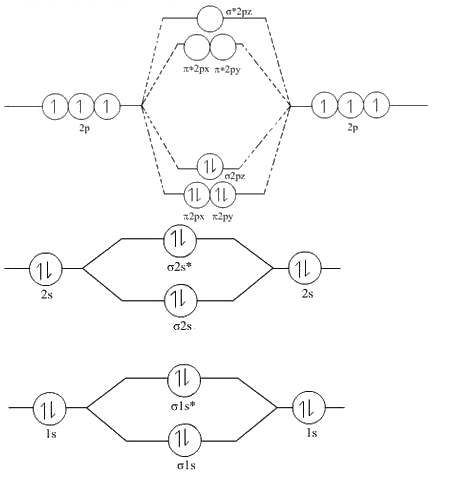
Explain Solution : M.O diagram shows that N2. There are a total of 17 electrons, 4 from each carbon and 1 from each hydrogen atom. PROBLEM 9-88 N2 has greater dissociation energy than N, whereas O2 has lower dissociation energy than O 2. The 2p x orbitals (one from each carbon atom) combine to form the three pi orbitals. The remaining four carbon 2sp 2 hybrid orbitals (one each on the end carbons and two on the middle carbon) combine to form 2 C-C bonding orbitals and 2 C-C antibonding orbitals. These levels are colored blue in the figure. The total, 11 electrons, fills orbitals from low energy to higher energy.Īllyl Radical In CH 2CHCH 2, 5 of the carbon 2sp 2 hybrid orbitals combine with the 5 hydrogen 1s orbitals to form 5 C-H bonding orbitals and 5 C-H antibonding orbitals. The nitrogen atom is less electronegative than the oxygen atoms so its atomic orbitals are a little higher in energy.īecause one of the non-bonding orbitals is localized on the N atom, it is a little higher in energy than the four non-bonding orbitals localized on the two oxygen atoms.īecause there is one 2p x orbital on each atom available for pi bonding, there are three pi symmetry molecular orbitals.Įach oxygen atom contributes is 6 valence electrons and the nitrogen contributes 5 electrons. Because of this, the highest energy orbital that is occupied by electrons, the 2 orbital, has only one electron.Īt right are the molecular orbitals for nitrogen dioxide. Nitrogen dioxide has one fewer electron than ozone. The full molecular orbital diagram with the electrons is below. 
3, antibonding, the mathematical sign of the wavefunction changes with every atom, repulsive interaction between atoms.2, non-bonding, zero pi electron density on the second atom.1, bonding all the way across the 3 atoms.The p orbitals in the picture above indicate electron density in those orbitals. The 2p x orbital on O 1, the 2p x orbital on O 2, and 2p x orbital on O 3 combine to form three pi symmetry orbitals. Two 2sp 2 orbitals on O 1, one 2sp 2 orbital on O 2, and two 2sp 2 orbitals on O 3 are non-bonding.O 3 uses one 2sp 2 orbital to combine with a second 2sp 2 orbital of O 2, making another sigma bonding and sigma antibonding orbital.O 1 uses one 2sp 2 orbital to combine with one 2p 2 orbital of O 2, making a sigma bonding and sigma antibonding orbital.Each oxygen atom combines its 2s, 2p z and 2p y orbitals to make three 2sp 2 hybrid orbitals. We'll use the hybrid orbital approximation. Let's look at the molecular orbital diagram of ozone. 
Therefore, Bond order = 1 / 2 Īs a result, the bond order of = 1 / 2 = 1 / 2 (4)=2 is 2.įor O 2 + molecule, an electron is removed from * 2 p y orbital.Pi Bonds over 3 Atoms Pi Bonds over 3 Atoms There are 10 bonding and 6 nonbonding electrons in the orbitals according to the molecular orbital configuration.Īccording to the molecular orbital configuration, there are 10 bonding and 6 nonbonding electrons in the orbitals. However, this picture is at odds with the magnetic behavior of oxygen. There is an OO double bond, and each oxygen atom has eight electrons around it. Energy level diagram for Molecular orbitals 1) Stability of molecules in terms of bonding and antibonding electrons 2) Stability of molecules in terms of bond. The molecule O 2 has the following molecular orbital configuration: We would write the following Lewis structure for O 2: This electronic structure adheres to all the rules governing Lewis theory. 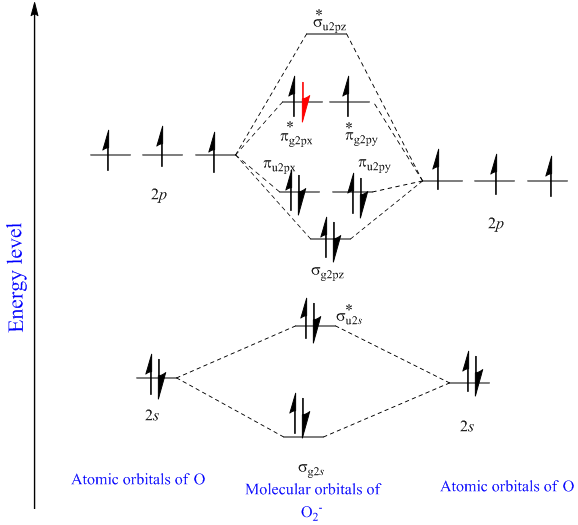
A total of 16 electrons make up an O 2 molecule. To begin, we can write the molecule’s molecular orbital arrangement. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
